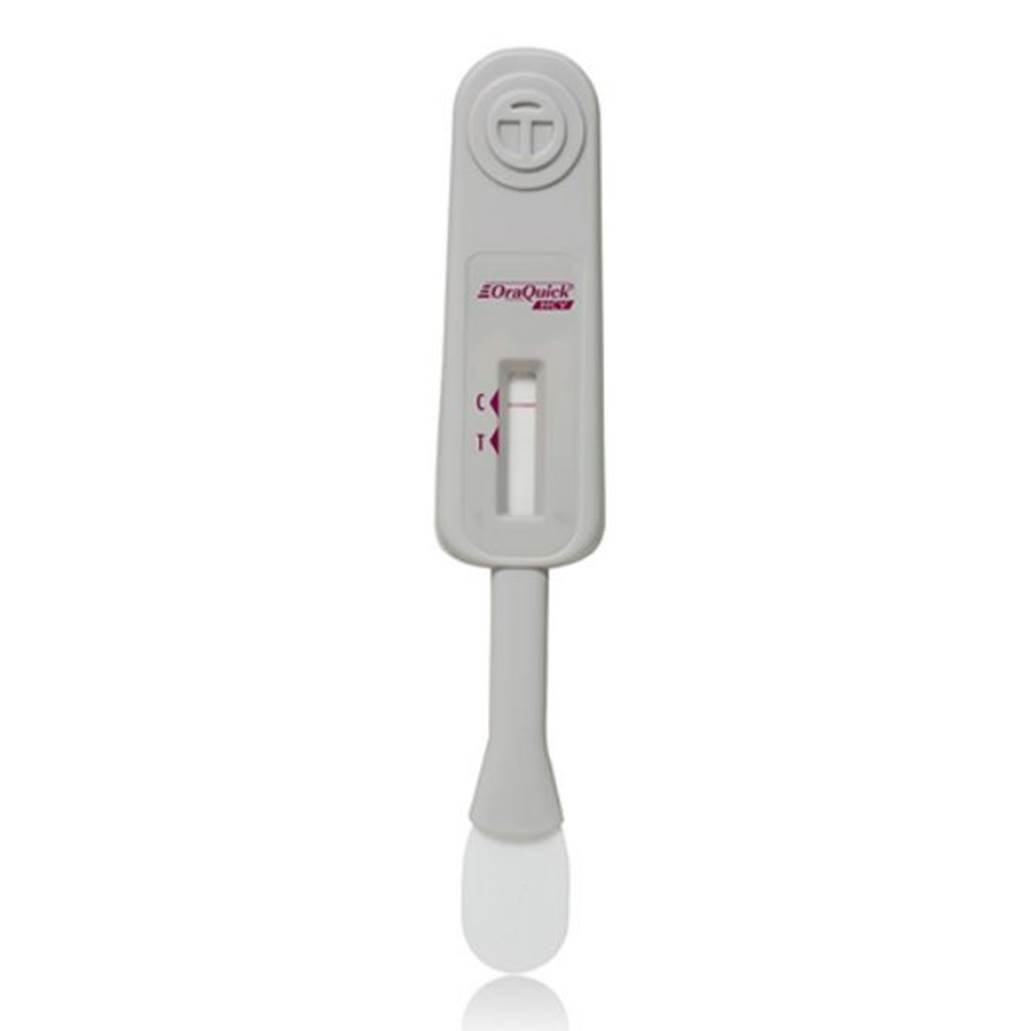
Product Description
The first FDA-approved, CLIA-waived rapid HCV test
The OraQuick® HCV test is FDA approved for detecting HCV antibodies in fingerstick and venipuncture whole blood. Our simple platform enables healthcare providers to deliver an accurate diagnosis in 20 minutes.
Accuracy
Clinically proven performance, OraQuick® HCV is greater than 98% accurate.
Appropriate For Testing In:
OraQuick® is CLIA-waived and is appropriate for testing in locations such as:
Public Health Settings, Physician Offices, Community-Health Clinics
Laboratories & Emergency Rooms
Features & Benefits
- Rapid Results: Point-of-care testing results in 20 minutes
- Laboratory Accuracy: Greater than 98% accurate and tests for multiple HCV genotypes
- Easy-to-Use: Fingerstick and venipuncture whole blood collection
- CLIA-waived
PRODUCT DETAILS
You may place order by giving us a call
Documentation and Videos
Coming Soon